
Leave message
Can’t find what you’re looking for?
Fill out this form to inquire about our custom protein services!
Inquire about our Custom Services >>



































| Cat. No. | Species | Product Description | Structure | Purity | Feature |
|---|---|---|---|---|---|
| FGB-H5259 | Human | Human FGF R3 (IIIb) / FGF R3B / CD333 Protein, Fc Tag (MALS verified) |  |

|
|

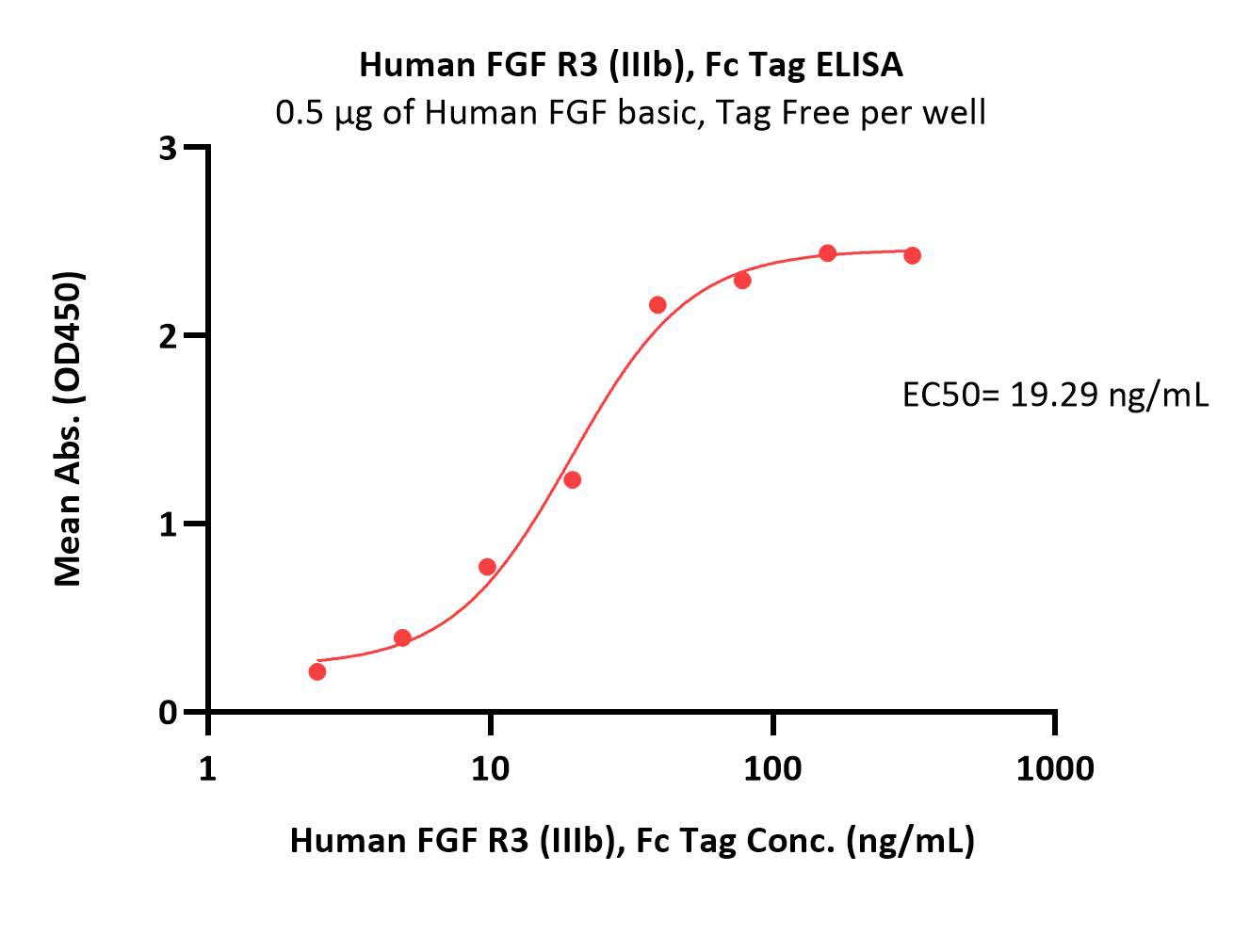
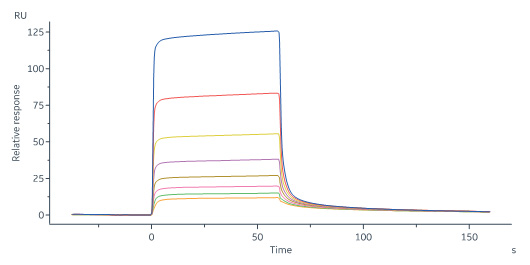
Human FGF R3 (IIIb), Fc Tag (Cat. No. FGB-H5259) immobilized on CM5 Chip can bind Human FGF acidic, Tag Free (Cat. No. AFF-H4116) with an affinity constant of 5.91 μM as determined in a SPR assay (Biacore 8K) (Routinely tested).
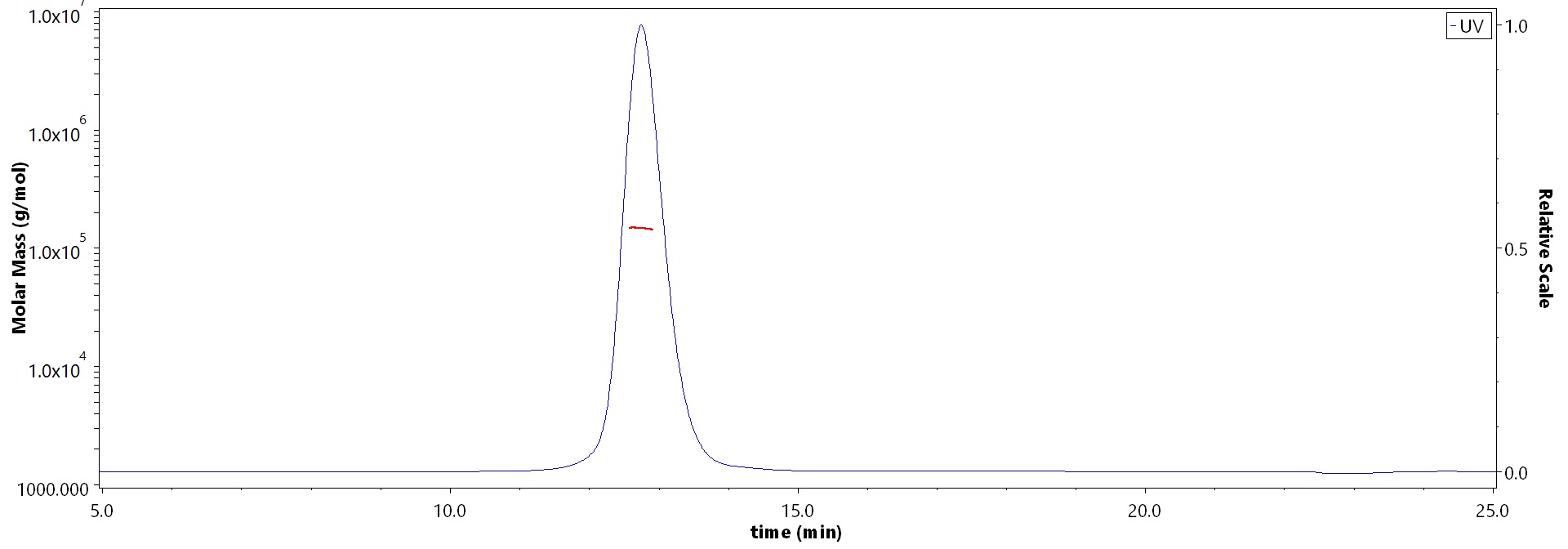
The purity of Human FGF R3 (IIIb), Fc Tag (Cat. No. FGB-H5259) is more than 90% and the molecular weight of this protein is around 132-162 kDa verified by SEC-MALS.
| Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Anlotinib Dihydrochloride | AL-3818 | Approved | Advenchen Laboratories Llc, Jiangsu Chia Tai-Tianqing Pharmaceutical Co Ltd | 福可维 | Mainland China | Carcinoma, Non-Small-Cell Lung | Jiangsu Chia Tai-Tianqing Pharmaceutical Co Ltd | 2018-05-08 | Thyroid Neoplasms; Peritoneal Neoplasms; Colorectal Neoplasms; Urologic Neoplasms; Gastrointestinal Stromal Tumors; Hepatic Insufficiency; Bone Neoplasms; Endometrial Neoplasms; Gallbladder Neoplasms; Fallopian Tube Neoplasms; Medullary thyroid cancer (MTC); Sarcoma, Alveolar Soft Part; Esophageal Squamous Cell Carcinoma; Carcinoma, Non-Small-Cell Lung; Uterine Cervical Neoplasms; Carcinoma, Hepatocellular; Melanoma; Osteoma; Gastroenteropancreatic neuroendocrine tumor; Small Cell Lung Carcinoma; Solid tumours; Ovarian Neoplasms; Head and Neck Neoplasms; Leiomyosarcoma; Esophageal Neoplasms; Stomach Neoplasms; Thoracic Neoplasms; Carcinoma, Renal Cell; Carcinoma, Ovarian Epithelial; Biliary Tract Neoplasms; Lymphoma, Large B-Cell, Diffuse; Neoplasms; Neuroendocrine Tumors; Sarcoma, Synovial; Liver Diseases; Sarcoma; Bile Duct Diseases; Nasopharyngeal Carcinoma | Details |
| Lenvatinib Mesylate | MK-7902; ER-203492-00; E-7080 | Approved | Eisai Co Ltd | Kisplyx, Lenvima, Lenvima/Kisplyx, 乐卫玛 | United States | Thyroid Neoplasms | Eisai Inc | 2015-02-13 | Neuroendocrine Tumors; Paraganglioma; Thyroid Cancer, Papillary; Melanoma; Neoplasm Metastasis; Carcinoma, Non-Small-Cell Lung; Carcinoma, Hepatocellular; Glioma; Lymphoma; Endometrial Neoplasms; Thyroid Neoplasms; Hepatic Insufficiency; Breast Neoplasms; Cholangiocarcinoma; Osteosarcoma; Solid tumours; Carcinoma, Adenoid Cystic; Adenocarcinoma, Follicular; Liver Diseases; Thyroid Carcinoma, Anaplastic; Adenocarcinoma of Lung; Neoplasms; Renal Insufficiency; Pheochromocytoma; Esophageal Neoplasms; Carcinoma, Renal Cell; Ovarian Neoplasms; Liver Neoplasms; Biliary Tract Neoplasms | Details |
| Nintedanib Esylate | BIBF-1120 | Approved | C.H. Boehringer Sohn Ag & Co. Kg | Ofev, Vargatef | United States | Idiopathic Pulmonary Fibrosis | Boehringer Ingelheim Gmbh | 2014-10-15 | Astrocytoma; Sarcoma; Prostatic Neoplasms; Breast Neoplasms; systemic sclerosis-associated interstitial lung disease; Adenocarcinoma, Clear Cell; Peritoneal Neoplasms; Colorectal Neoplasms; Silicosis; Genital Neoplasms, Female; Hepatic Insufficiency; Oligodendroglioma; Gliosarcoma; Uterine Neoplasms; Endometrial Neoplasms; Fallopian Tube Neoplasms; Carcinoma, Squamous Cell; Leukemia, Myeloid, Acute; Appendiceal Neoplasms; Carcinoma, Non-Small-Cell Lung; Carcinoma, Endometrioid; Carcinoma, Hepatocellular; Glioblastoma; Solid tumours; Telangiectasia, Hereditary Hemorrhagic; Carcinoma, Renal Cell; Endometrial Stromal Tumors; Carcinoid Tumor; Radiation Pneumonitis; Esophageal Neoplasms; Rejection of lung transplantation; Idiopathic Pulmonary Fibrosis; Neoplasms; Ovarian Neoplasms; Small Cell Lung Carcinoma; Colonic Neoplasms; Scleroderma, Systemic; Lung Diseases, Interstitial; Pulmonary Fibrosis; Mesothelioma; Multiple Myeloma; Asbestosis; Neuroendocrine Tumors | Details |
| Futibatinib | TAS-120 | Approved | Taiho Pharmaceutical Co Ltd, Otsuka Pharmaceutical Co Ltd | LYTGOBI | United States | Cholangiocarcinoma | Taiho Oncology Inc | 2022-09-30 | Central Nervous System Neoplasms; Carcinoma, Non-Small-Cell Lung; Neoplasm Metastasis; Endometrial Neoplasms; Lymphoma; Metastatic breast cancer; Sarcoma; Cholangiocarcinoma; Breast Neoplasms; Biliary Tract Neoplasms; Carcinoma, Transitional Cell; Neoplasms; Esophageal Neoplasms; Carcinoma; Stomach Neoplasms; Bone Marrow Neoplasms; Solid tumours | Details |
| Selpercatinib | ARRY-192; Ret-IN-1; LOXO-292; LY-3527723 | Approved | Array Biopharma | Retevmo, Retsevmo | United States | Medullary thyroid cancer (MTC); Carcinoma, Non-Small-Cell Lung; Thyroid Neoplasms | Loxo Oncology Inc | 2020-05-08 | Medullary thyroid cancer (MTC); Neuroectodermal Tumors, Primitive, Peripheral; Neoplasms, Germ Cell and Embryonal; Thyroid Cancer, Papillary; Carcinoma, Non-Small-Cell Lung; Glioma; Lung Neoplasms; Lymphoma; Lymphoma, Non-Hodgkin; Carcinoma, Squamous Cell; Thyroid Neoplasms; Hepatic Insufficiency; Neuroblastoma; Histiocytosis, Langerhans-Cell; Sarcoma, Ewing; Osteosarcoma; Fibrosarcoma; Sarcoma; Myofibromatosis; Wilms Tumor; Neoplasms; Thyroid Carcinoma, Anaplastic; Colonic Neoplasms; Hepatoblastoma; Rhabdoid Tumor; Renal Insufficiency; Medulloblastoma; Ependymoma; Rhabdomyosarcoma; Hematologic Neoplasms; Solid tumours | Details |
| Pemigatinib | INCB-054828; INCB-54828; IBI-375 | Approved | Incyte Corp | Pemazyre, 伯坦, 达伯坦 | United States | Cholangiocarcinoma | Incyte Corp | 2020-04-17 | Cholangiocarcinoma; Carcinoma, Non-Small-Cell Lung; Adenocarcinoma; Lymphoma, Non-Hodgkin; Glioma; Endometrial Neoplasms; Lung Neoplasms; Lymphoma; Urologic Neoplasms; Colorectal Neoplasms; Bile Duct Neoplasms; Colitis, Ulcerative; Translocation, Genetic; Breast Neoplasms; Biliary Tract Neoplasms; Urinary Bladder Neoplasms; Multiple Myeloma; Neoplasms; Pancreatic Neoplasms; Glioblastoma; Carcinoma, Transitional Cell; Myeloproliferative Disorders; Carcinoma, Renal Cell; Carcinoma; Stomach Neoplasms; Solid tumours; Bone Marrow Neoplasms | Details |
| Pazopanib Hydrochloride | GSK-786034; GW-786034B; SB-786034; GW-786034 | Approved | Glaxosmithkline Plc, Novartis Pharma Ag | 维全特, Armala, Votrient, Patorma | United States | Carcinoma, Renal Cell; Sarcoma | Novartis Pharma Ag | 2009-10-19 | Osteosarcoma; Germinoma; Fallopian Tube Neoplasms; Leukemia, Myeloid, Acute; Colorectal Neoplasms; Uterine Cervical Diseases; Genital Neoplasms, Female; Gliosarcoma; Gastrointestinal Stromal Tumors; Peritoneal Neoplasms; Chondrosarcoma, Extraskeletal Myxoid; Lung Neoplasms; Nasopharyngeal Carcinoma; Medullary thyroid cancer (MTC); Brain Neoplasms; Urethral Neoplasms; Psoriasis; Gastrinoma; Neuroblastoma; Prostatic Neoplasms; Breast Neoplasms; Neoplasms, Germ Cell and Embryonal; von Hippel-Lindau Disease; Thyroid Cancer, Papillary; Neoplasm Metastasis; Breast Neoplasms, Male; Uterine Cervical Neoplasms; Endodermal Sinus Tumor; Paraganglioma; Melanoma; Macular Degeneration; Carcinoma, Hepatocellular; Sarcoma; Gastrointestinal Neoplasms; Carcinoma, Non-Small-Cell Lung; Thyroid Neoplasms; Choriocarcinoma; Uterine Neoplasms; Lymphoma; Carcinoma, Neuroendocrine; Glioma; Carcinoma, Embryonal; Skin Melanoma; Neoplasms; Herpes Genitalis; Hemangiosarcoma; Squamous Cell Carcinoma of Head and Neck; Insulinoma; Pheochromo | Details |
| Tasurgratinib | E-7090 | Approved | Eisai Co Ltd | TASFYGO | Japan | Biliary Tract Neoplasms | Eisai Co Ltd | 2024-09-24 | Biliary Tract Neoplasms; Solid tumours; Neoplasms; Cholangiocarcinoma; Breast Neoplasms; Hepatic Insufficiency | Details |
| Erdafitinib | 890E37NHMV; G-024; JNJ-493; JNJ-42756493; TAR-210 | Approved | Astex Pharmaceuticals Inc | Balversa, 博珂 | United States | Carcinoma, Transitional Cell | Janssen Biotech Inc | 2019-04-12 | Carcinoma, Squamous Cell; Osteosarcoma; Sarcoma, Ewing; Neuroblastoma; Prostatic Neoplasms; Bile Duct Neoplasms; Urologic Neoplasms; Histiocytic Sarcoma; Hepatic Insufficiency; Lymphoma; Lymphoma, Non-Hodgkin; Sarcoma; Glioma; Metastatic breast cancer; Carcinoma, Neuroendocrine; Carcinoma, Hepatocellular; Carcinoma, Non-Small-Cell Lung; Neoplasms, Germ Cell and Embryonal; Neuroectodermal Tumors, Primitive, Peripheral; Xanthogranuloma, Juvenile; Adenocarcinoma; Hepatoblastoma; Ependymoma; Medulloblastoma; Rhabdomyosarcoma; Hematologic Neoplasms; Bone metastases; Esophageal Neoplasms; Stomach Neoplasms; Carcinoma; Rhabdoid Tumor; Solid tumours; Glioblastoma; Carcinoma, Transitional Cell; Neoplasms; Wilms Tumor; Multiple Myeloma; Urinary Bladder Neoplasms; Central Nervous System Neoplasms; Prostatic Neoplasms, Castration-Resistant; Histiocytosis, Langerhans-Cell | Details |
| Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| Rogaratinib | BAY-1163877 | Phase 3 Clinical | Bayer AG | Solid tumours; Neoplasms; Carcinoma, Transitional Cell; Urinary Bladder Neoplasms; Breast Neoplasms; Sarcoma; Gastrointestinal Stromal Tumors; Metastatic breast cancer; Carcinoma, Non-Small-Cell Lung | Details |
| Infigratinib | BGJ-398; NVP-BGJ398 | Phase 3 Clinical | Novartis Pharma Ag | Pancreatic Neoplasms; Uterine Cervical Neoplasms; Carcinoma, Pancreatic Ductal; Carcinoma, Squamous Cell; Glioma; Gastrointestinal Stromal Tumors; Oropharyngeal Neoplasms; Breast Neoplasms; Cholangiocarcinoma; Central Nervous System Neoplasms; Urinary Bladder Neoplasms; Carcinoma, Transitional Cell; Colonic Neoplasms; Biliary Tract Neoplasms; Glioblastoma; Achondroplasia; Papillomavirus Infections; Neoplasms; Nasopharyngeal Neoplasms; Rectal Neoplasms; Squamous Cell Carcinoma of Head and Neck; Stomach Neoplasms; Esophageal Neoplasms; Hematologic Neoplasms; Solid tumours | Details |
| SC-0011 | SC-0011 | Phase 3 Clinical | Shijiazhuang Zhikang Hongren New Drug Development Co Ltd | Solid tumours; Esophageal Neoplasms; Stomach Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| Zoligratinib | CH-5183284; FF-284; Debio-1347 | Phase 2 Clinical | Chugai Pharmaceutical Co Ltd | Solid tumours; Breast Neoplasms | Details |
| AUR-109 | ODM-203; AUR-109 | Phase 2 Clinical | Orion Corp | Ovarian Neoplasms; Liver Neoplasms; Kidney Neoplasms; Solid tumours; Carcinoma, Renal Cell; Urinary Bladder Neoplasms; Pulmonary Fibrosis; Breast Neoplasms; Colorectal Neoplasms; Lung Neoplasms | Details |
| Lucitanib | S-80881; AL-3810; CO-3810; E-3810; S-80881-2 | Phase 2 Clinical | Advenchen Laboratories Nanjing Ltd | Solid tumours; Stomach Neoplasms; Small Cell Lung Carcinoma; Breast Neoplasms; Nasopharyngeal Carcinoma; Genital Neoplasms, Female; Colorectal Neoplasms; Thymus Neoplasms; Lung Neoplasms; Metastatic breast cancer; Carcinoma, Non-Small-Cell Lung; Carcinoma, Hepatocellular | Details |
| Fexagratinib | AZD4547; AZD-4547; ABSK-091; ABSK091 | Phase 2 Clinical | Astrazeneca Plc | Lymphoma; Multiple Myeloma; Breast Neoplasms; Prostatic Neoplasms; Colorectal Neoplasms; Carcinoma, Squamous Cell; Lung Neoplasms; Glioma; Uterine Neoplasms; Urinary Bladder Neoplasms; Thyroid Neoplasms; Endometrial Neoplasms; Metastatic breast cancer; Melanoma; Carcinoma, Non-Small-Cell Lung; Foodborne Diseases; Adenocarcinoma; Uterine Cervical Neoplasms; Rectal Neoplasms; Solid tumours; Ovarian Neoplasms; Head and Neck Neoplasms; Hematologic Neoplasms; Stomach Neoplasms; Gastrointestinal Diseases; Squamous Cell Carcinoma of Head and Neck; Esophageal Neoplasms; Liver Neoplasms; Carcinoma, Renal Cell; Neoplasms; Carcinoma, Transitional Cell; Colonic Neoplasms; Carcinoma, Ovarian Epithelial; Pancreatic Neoplasms; Skin Neoplasms | Details |
| Derazantinib | ARQ-087.2HCl; AQ-14741087; ARQ-087; BAL-087 | Phase 2 Clinical | Arqule Inc | Solid tumours; Biliary Tract Neoplasms; Stomach Neoplasms; Carcinoma, Transitional Cell; Cholangiocarcinoma; Urogenital Neoplasms; Carcinoma, Hepatocellular | Details |
| HH185 | 3D-185; 3-D185; HH-185; 3D185 | Phase 2 Clinical | ShangHai HaiHe Biopharma Co Ltd, Shanghai Institute Of Materia Medica, Chinese Academy Of Sciences, Shanghai Medicilon Inc | Solid tumours; Cholangiocarcinoma | Details |
| TYRA-300 | TYRA300 | Phase 2 Clinical | Tyra Biosciences Inc | Solid tumours; Carcinoma; Carcinoma, Transitional Cell; Achondroplasia; Urinary Bladder Neoplasms; Urologic Neoplasms | Details |
| SAR-442501 | SAR-442501 | Phase 2 Clinical | Sanofi | Osteochondrodysplasias; Achondroplasia | Details |
| HMPL-453 | HMPL-453 | Phase 2 Clinical | Hutchison Medipharma Ltd | Solid tumours; Biliary Tract Neoplasms; Neoplasms, Mesothelial; Mesothelioma; Cholangiocarcinoma | Details |
| ABSK-061 | ABSK061 | Phase 2 Clinical | ABbisko Therapeutics Co Ltd | Solid tumours; Esophageal Neoplasms; Carcinoma, Transitional Cell; Carcinoma, Non-Small-Cell Lung | Details |
| PZH-2111 | PZH2111; PZH-2111 | Phase 2 Clinical | Zhangzhou Pientzehuang Pharmaceutical Co Ltd | Solid tumours; Cholangiocarcinoma | Details |
| Gunagratinib | ICP-192 | Phase 2 Clinical | Beijing Tiancheng Pharmaceutical Technology Co Ltd | Solid tumours; Biliary Tract Neoplasms; Head and Neck Neoplasms; Stomach Neoplasms; Carcinoma, Transitional Cell; Urinary Bladder Neoplasms; Cholangiocarcinoma; Lung Neoplasms | Details |
| Recifercept | PF-07256472; TA-46 | Phase 1 Clinical | Pfizer Inc | Achondroplasia | Details |
| BPI-17509 | BPI-17509 | Phase 1 Clinical | Betta Pharmaceuticals Co Ltd | Solid tumours | Details |
| nintedanib (Avalyn Pharma) | Phase 1 Clinical | Avalyn Pharma Inc | Idiopathic Pulmonary Fibrosis; Pulmonary Fibrosis | Details | |
| TYRA-200 | TYRA200; TYRA-200 | Phase 1 Clinical | Tyra Biosciences Inc | Solid tumours; Biliary Tract Neoplasms; Cholangiocarcinoma; Bile Duct Neoplasms | Details |
| Resigratinib | KIN-3248; KIN-003 | Phase 1 Clinical | Kinnate Biopharma Inc | Solid tumours; Carcinoma, Transitional Cell; Neoplasms; Cholangiocarcinoma | Details |
| MNKD-201 | MNKD-201; MKND-201 | Phase 1 Clinical | MannKind Corp | Idiopathic Pulmonary Fibrosis | Details |
| Inhaled Nintedanib – dry powder (Avalyn Pharma) | AP02; AP 02 DP; AP-02; AP02-DP | Phase 1 Clinical | Avalyn Pharma Inc | Idiopathic Pulmonary Fibrosis; Respiratory Tract Diseases; Lung Diseases, Interstitial; Pulmonary Fibrosis | Details |
| TYRA-300-B01 | TYRA-300-B01 | Phase 1 Clinical | Tyra Biosciences Inc | Details | |
| LOXO-435 | LOXO-435; LY3866288; LY-3866288; [14C]-LY3866288 | Phase 1 Clinical | Eli Lilly And Company | Solid tumours; Carcinoma, Transitional Cell; Neoplasms; Urinary Bladder Neoplasms; Ureteral Neoplasms; Neoplasm Metastasis | Details |
| GR-1017 | GR1017; GR-1017; AMB302; AMB-302; GQ1011; GQ-1011 | Phase 1 Clinical | Aimed Bio Inc, GeneQuantum Healthcare (Suzhou) Co Ltd | Solid tumours; Glioblastoma; Urinary Bladder Neoplasms | Details |
| TT-00434 | TT-00434 | Phase 1 Clinical | TransThera Sciences (Nanjing) Inc | Solid tumours; Urinary Bladder Neoplasms | Details |
| ABSK-121 | ABSK121-NX; ABSK121 | Phase 1 Clinical | ABbisko Therapeutics Co Ltd | Solid tumours | Details |
| LY-2874455 | LY-2874455 | Phase 1 Clinical | Eli Lilly And Company | Neoplasms; Leukemia, Myeloid, Acute | Details |
| CPL-304110 | CPL-304110; CPL-304-110 | Phase 1 Clinical | Celon Pharma Sa | Stomach Neoplasms; Urinary Bladder Neoplasms; Cholangiocarcinoma; Sarcoma; Endometrial Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
This web search service is supported by Google Inc.







